Toxicokinetics Evaluation
Toxicokinetic (TK) evaluation is very useful in understanding the connection between toxicity and administered dose. As an integrated CRO, our company has extensive expertise in TK evaluation. We employ talented and highly trained scientists who focus on every aspect of rare disease therapy research and development and can customize experimental designs for your project according to your specific requirements.
Overview of Toxicokinetics Evaluation
Toxicological (TK) evaluation is a branch of pharmacological evaluation and is used to study the dynamic changes of toxicity test doses in the body and obtain pharmacokinetic data. PK evaluation can obtain sufficient information on drug absorption, distribution, metabolism, and excretion to correlate toxicity findings with appropriate levels of drug exposure and facilitate the study of toxicity mechanisms.
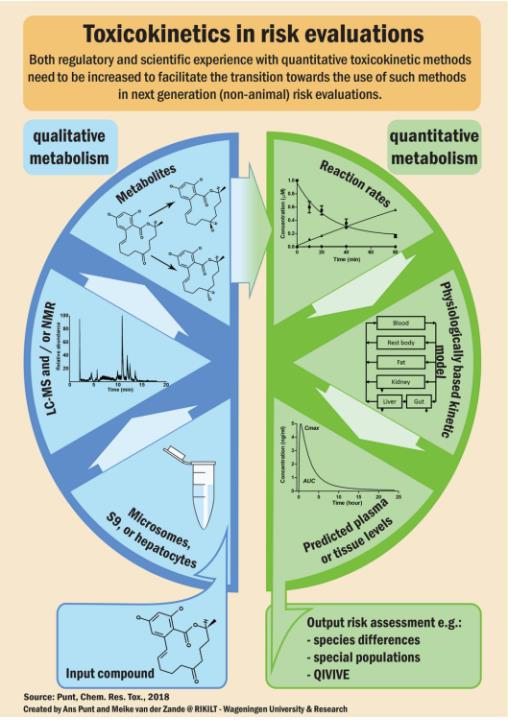 Fig.1 Diagram of toxicokinetics in risk evaluations. (Punt, Ans, 2018)
Fig.1 Diagram of toxicokinetics in risk evaluations. (Punt, Ans, 2018) Significance of Toxicokinetics Evaluation
- Describe the systemic exposure of the compound to animals and its relationship to toxicity study dose levels and toxicity study periods.
- Understand the relationship between drug exposure and toxicology results.
- Support animal species selection and dosing regimens for nonclinical toxicity studies.
- Combined with toxicity study results, provide information useful for subsequent non-clinical toxicity studies.
Differences Between Toxicokinetics (TK) and Pharmacokinetics (PK)
Toxicokinetic (TK) evaluation is similar to pharmacokinetic (PK) evaluation, but there are some differences.
| Toxicokinetic (TK) | Pharmacokinetic (PK) | |
|---|---|---|
| Study Dose | High dose | Dose escalation |
| Study Objects | Animals | Animals and humans |
| Study Purposes | Determining the exposure levels at which toxic effects are observed and using the results to guide dosing recommendations for clinical trials. | Understanding drug clearance and bioavailability in biological systems to improve drug efficacy. |
Our Services
With years of continuous improvement and the construction of animal platforms, our company can provide mice, rats, dogs, pigs, and non-human primates for TK evaluation. Our expert team has extensive experience in TK study design and can provide services such as dose selection, frequency, and sample collections according to customer requirements.
Services we can provide include but are not limited to:
- TK support for toxicology studies
- Dose correlation analysis
- Specific parameters analysis (Cmax, Tmax, AUC, t1/2, clearance (CL), volume of distribution (VD), etc.
- TK/TOX analysis
- Metabolite analysis
- Immunogenicity analysis
- Microsampling for TK profiling
In addition, our advanced platform technologies, such as LC-MS/MS and ELISA, are available to support TK analysis. We have the capabilities and resources to provide end-to-end TK evaluation services to facilitate the development of your programs.
Why Choose Us?

- Customized services and products tailored to your research
- One-stop platform with an experienced technical team
- The rich experience accumulated in successful cases of new drug development
- High data quality and reliable analysis
- Exceptional post-sales support
Project Workflow

With a research team with extensive expertise in drug safety evaluation, our company is confident to provide clients with toxicokinetics evaluation services for rare diseases. We provide professional communication and problem-solving support to ensure that we can quickly respond to the changing needs of your rare disease therapy research projects. If you are interested in our services, please feel free to contact us for more information.
Reference
- Punt, Ans. "Toxicokinetics in risk evaluations." Chemical Research in Toxicology 31.5 (2018): 285-286.
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.
